Foxglove Aphids in Lettuce: Control with Reduced-Risk
and Conventional Insecticides
John C. Palumbo
Note: Foxglove aphids have been observed colonizing head and leaf lettuce in experimental plots at the Yuma Agricultural Center since mid-January. The intensity of observed infestation levels are similar to what was observed this time last year. PCA’s should be aware of the presence of this aphid species, particularly when temperatures begin to warm up. For assistance in identification of foxglove aphids visit: http://ag.arizona.edu/crops/vegetables/insects/advisories/2003/JCPaphidkey1203.pdf or contact us at the Agricultural Center.
Introduction
As a key pest of desert lettuce, aphids represent one of the most important
insect problems currently facing the industry. A new aphid species, the
foxglove aphid, Aulacorthum solani, was found infesting commercial
lettuce fields in the Yuma area for the first time this past growing season.
It has been present in California since at least 1940, and has caused
problems for lettuce growers in Salinas for the past 5 years. The foxglove
aphid was first discovered infesting head lettuce at low levels the Yuma
Agricultural Center in the spring of 2001. Then in the spring of 2002
foxglove aphids reached high population levels at YAC on spring plantings,
but were not reported in commercial fields. This past spring foxglove
aphid populations were wide-spread throughout the Yuma Valley, particularly
in fields near the Colorado river and adjacent to citrus orchards and
residential areas.
Unlike the lettuce aphid which was first found in Yuma five years ago, the foxglove aphid is known to colonize a much broader range of plant hosts, including a wide variety of weeds (i.e., Shepards purse, ground cherry, pigweed), ornamentals (i.e., geraniums, gladiolas, verbena) and crops (i.e., cucurbits, beans, canola, spinach, citrus, safflower, tomatoes). This large availability of hosts and apparent adaptation to our winter and spring growing conditions suggests that foxglove aphids might present growers with some new challenges.
Although we are uncertain how this new species will behave under desert growing conditions in the long-term, infestations this spring reached high levels in experimental plots and in some commercial fields. Another aphid species, Acyrthosiphon lactucae (no common name) which is commonly misidentified as potato aphid, was also present this year in extremely high levels. Because of the importance of these aphids as contaminants of lettuce and other leafy vegetables, we designed several insecticide trials this past season to determine how effective conventional, reduced-risk and other new insecticides were against these aphids under local growing conditions
Materials and Methods
Small-plot, field studies were conducted in head lettuce and romaine at the University of Arizona, Yuma Agricultural Center in the spring 2003 growing seasons to evaluate the efficacy of several new reduced risk and conventional insecticides for control of aphids and thrips. In each trial, lettuce was direct seeded into double row beds on 42 inch centers and sprinkled beginning the following day. Plots for each trial consisted of 2-4 beds, 50' long with a two bed buffer between the plots. Plots were arranged in a randomized complete block design with 4 replications. Treatments and rates for each crop are presented in the data tables. Specific information for each trial is listed below:
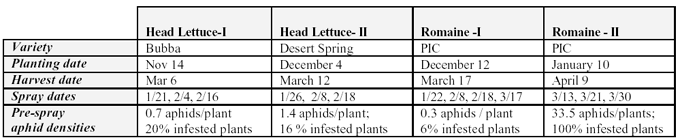
In the head lettuce-I trial, the at-planting soil applications of Admire, Platinum and dinotefuron were applied as a preplant injection at a depth of 1.5" below the seed line at bed shaping in 15 GPA final dilution. The side dress treatment of Platinum was applied at 2nd side dress (Jan 15) similar to fertilizer side dressing and the materials were placed on the bed shoulder at @ 3” below the soil in 30 GPA final dilution. In all trials, foliar spray applications were hand applied with a CO2 operated boom sprayer operated at 60 psi and 27 GPA. A directed spray (~75% band, with rate adjusted for band; nozzles directed inward toward the plants) was delivered through 3 nozzles (TX-10) per bed. An adjuvant was applied to all foliar treatments; DyneAmic, Exit, Hook or Induce at 0.065%v/v. The first spray in each trial was initiated when aphids were first found colonizing plants (see above; pre-spray aphid densities).
Aphid populations were assessed by estimating the number of aphids /plant in whole plant, destructive samples. Five aphid species were present on plants during the trials and have been classified into two groups for analysis in the following (1) Foxglove aphid, and (2) Green aphid complex consisting of Acyrthosiphon lactucae (no common name), potato aphid and green peach aphid. In addition, Lettuce aphids were present in significant numbers during the Romaine-II trial. On each sampling date, 8-10 plants were randomly selected from each plot and placed individually into large 5-gal tubs. Each plant was sampled by visually examining all plant foliage and counting the number of alate (winged) and apterous (non-winged) aphids present. In the head lettuce, infestation levels of apterous aphids at harvest were estimated by randomly selecting 10 plants within each replicate, visually counting the number of aphids on frame/wrap per leaves and heads separately. In romaine, all leaves were sampled when plants were harvested and particular attention was given to hearts and terminal growing points.
Results and Discussion
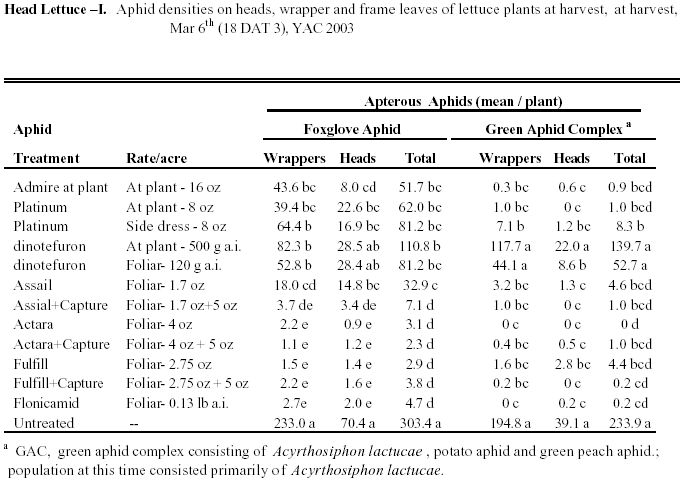
Head Lettuce I: Foxglove aphids were the dominant aphid
species during this trial, but A. lactucae was also very abundant
(Table 1). Several new insecticides, both soil and
foliar applied, were evaluated for economic aphid control. The soil applied
neonicotinoid treatments provided marginal control of foxglove aphids
at harvest, and head contamination was lowest in the Admire (16 oz) plots
(Table 1). None of the soil treatments were comparable
to the foliar sprays in controlling foxglove aphids, but all of the soil
treatments except dinotefuron suppressed populations of the green aphid
complex to very low levels. In most cases, all of the foliars reduced
aphid numbers to acceptable levels at harvest. Dinotefuron did not provided
significant control relative to the other foliar compounds, and Assail
applied alone, provided marginal control of Foxglove aphid. When combined
with Capture, Assail provided significantly better control. Flonicamid,
Actara and Fulfill provided excellent control of all aphids, and addition
of Capture did not significantly improve efficacy.
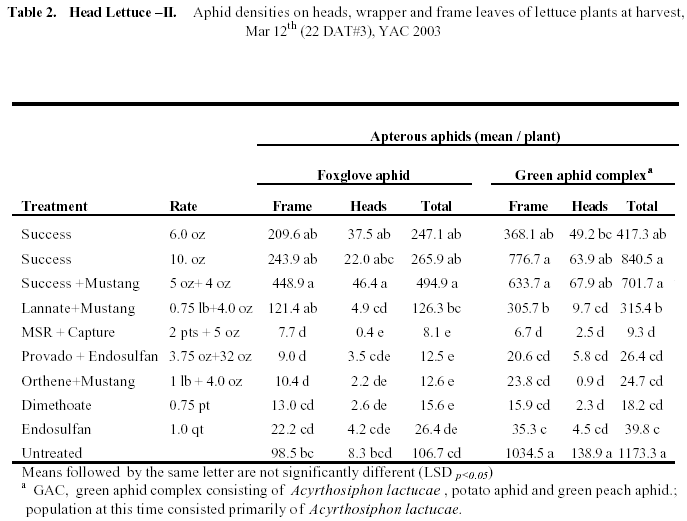
Head Lettuce II: Foxglove aphid abundance was lower in this trial, whereas A. lactucae abundance was extremely high (Table 2). Interestingly, green peach and potato aphid populations were low, comprising less than 10% of the green aphid complex. This trial was originally designed to evaluate thrips (thus the inclusion of the Success treatments) but we focused on the aphids due to their overwhelming presence in the plots. All treatments were applied as foliar spray and the 3rd application was made 22 days before harvest. The Success treatments did not control aphids and in fact actually resulted in greater numbers of foxglove aphids than the untreated control. The Lannate + Mustang treatments did significantly suppress Foxglove aphid numbers, but did not significantly lower numbers of green aphids compared with the untreated check. All of the older compounds evaluated provided significant control of all aphid species at harvest (>95% control). In particular, MSR+Capture, Othehene+Mustang and dimethoate provided very good residual efficacy. Endosulfan applied alone was a little less consistent on Foxglove aphid.
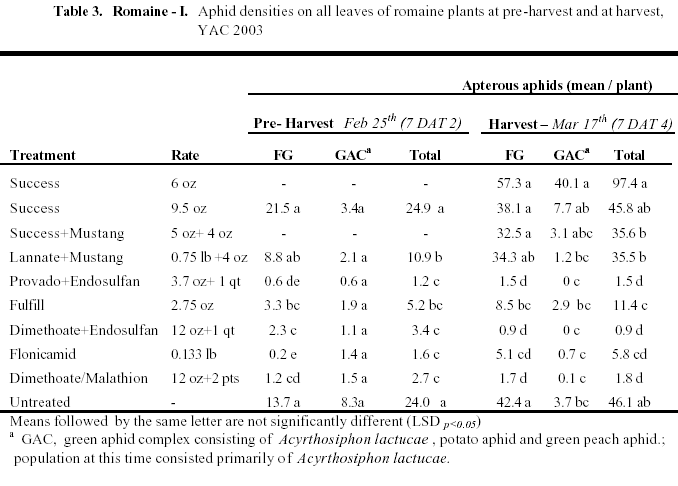
Romaine I: This trial was designed similar to the above trial in head lettuce, but only older treatments registered on Romaine were evaluated. Aphid numbers in general were lower in this trial, not exceeding 100 aphids/plant (Table 3). Foxglove aphids were the dominant aphid species in the untreated plots, but GAC were actually higher in the Success (6 oz) treatment than in the check. Similar to the above trials, Success and Lannate did not significantly reduce aphid numbers. Foxglove aphids were best controlled in plots treated with dimethoate, Provado, endosulfan, and Flonicamid. Overall, the dimethoate and endosulfan combination was particularly effective against aphids during the trial. Fulfill did reduce numbers at significantly lower levels than the check, but not as efficaciously as the older compounds.
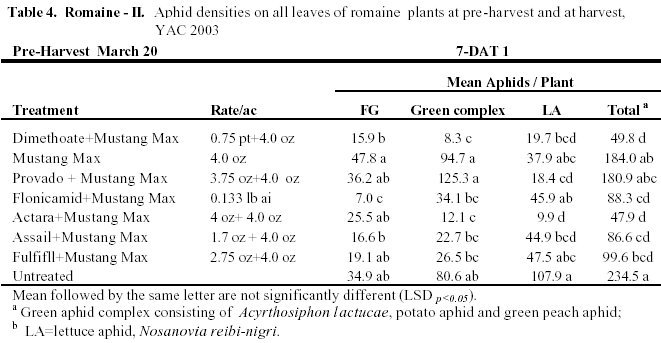
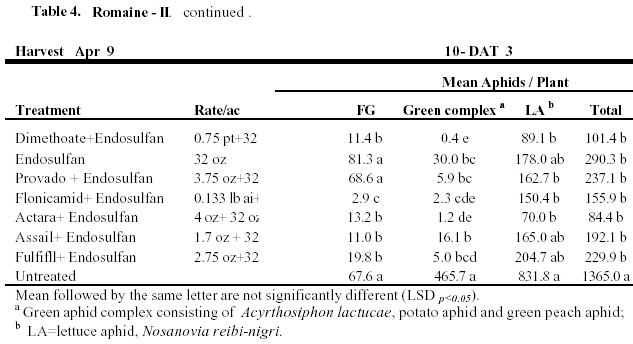
Romaine II: This test was a late season trial on romaine where aphid numbers were allowed to establish on plants prior to spray initiation. Numbers of aphids exceeded 33 aphids / plant before the 1st application where lettuce aphids were 15.5 aphids/ plant, green aphids were about 12 aphids/plant and foxglove were 5.1 aphids. We also focused on tank mixtures with older contact materials and newer systemic compounds to evaluate both knockdown and residual control. For the 1st spray all treatments were combined with Mustang. Flonicamid provided the most significant efficacy of Foxglove at 7-DAT, but was not as effective against lettuce aphids (Table 4). The dimethoate and Actara treatments provided the most significant knockdown efficacy against Green and Lettuce aphids. Following the 2nd spray combined with Capture, again flonicamid provided the best foxglove aphid control. Dimethoate provided good green and lettuce aphid knockdown, but appeared to be less effective on foxglove aphid. Relative to the untreated check, Actara provided good efficacy of both green and lettuce aphids. Residual efficacy of foxglove aphid following the 3rd spray was again most significant with the Flonicamid combination. Similar to the previous application, Actara and dimethoate provided good sustained knockdown of the green aphid complex. Although, both of these treatments provided >90% control of lettuce aphids at 10 DAT #3, romaine hearts harvested in these plots were not marketable due to excessive aphid infestation (>70 aphids/heart).
Conclusions and Recommendations
• Conventional Insecticides: Dimethoate, MSR and Orthene consistently provided economic efficacy of foxglove and green aphids in head lettuce and romaine. These products should be used at high rates and users should be aware of the restrictive PHI and REI for each compound.
• Reduced-risk Insecticides: Flonicamid and Actara consistently provided good systemic residual control of foxglove aphids and the green aphid complex. Fulfill was less consistent, but provided good residual control on head lettuce when applied to low aphid densities. Assail provided inconsistent efficacy and performed best when combined with a contact insecticide. Presently, Actara, Flonicamid and Assail are not registered for use on lettuce in Arizona, but labels may be available in 1-2 years. Based on field performance to date, these products should be used at higher rates.
• Soil-applied Insecticides: Admire appeared to provide less control of Foxglove aphids (@ 85-90% control) than green aphids (>95%) in head lettuce. However, other studies this past spring showed that Admire provided good control of foxglove aphids (90+ % control), particularly when populations were lighter. At best, Platinum appears to be comparable to Admire, and dinotefuron has shown inconsistent performance as both a soil and foliar treatment.
• Sampling: Whole plant samples should be taken season long, with particular attention paid to the lower frame leaves for the green aphid complex and foxglove aphids, and the terminal growth for lettuce aphids. When alatae (winged forms) are found on plants, they should be keyed out to species. Many non-pest alates aphids can be found on lettuce but will not significantly colonize (i.e. cowpea aphid, cabbage aphid, english grain aphid). However, presence of alatae foxglove and green complex aphids is usually followed by colonization by apterous forms (colonizing non winged). Sampling should then be intensified to detect colonization.
• Action Threshold: The compounds used in these trials should be applied when aphids first begin to colonize. This is consistent with a threshold level of 5-10% infested plants (the percentage of plants infested with 1 or more non-winged aphids). Sprays should be initiated at when this level is exceeded and preventing colonization at harvest has been most consistent using spray intervals of 14-18 days. None of the products tested have the capability to rapidly knockdown heavy aphid infestations on heading plants.
Acknowledgments
Funding and support for this research was provided by the following sources:
the Arizona Iceberg Lettuce Research Council, Bayer Crop Science, Syngenta
Crop Protection, FMC Corp., and Valent USA Corp. Special thanks to Todd
Hannan, Gowan Seed Co., for providing seed used in some of these studies.
I gratefully acknowledge the excellent assistance from the personnel at
the Yuma Agricultural Center including Clay Mullis, Andreas Amaya, Luis
Ledesma, Leonardo Chavez, Javier Ruiz and Gerardo Villegas.

Issued in furtherance of Cooperative Extension work, acts of May 8 and June 30, 1914, in cooperation with the U.S. Department of Agriculture, James A. Christenson, Director Cooperative Extension, College of Agriculture and Life Sciences, The University of Arizona.
The University of Arizona is an equal opportunity, affirmative action institution. The University does not discriminate on the basis of race, color, religion, sex, national origin, age, disability, veteran status, or sexual orientation in its programs and activities.
Any products, services, or organizations that are
mentioned, shown, or indirectly implied in this web document do not imply
endorsement by The University of Arizona.
Information provided by:
John C. Palumbo, jpalumbo@ag.arizona.edu Research Scientist (Entomology)
University of Arizona, Tucson, Arizona.
Material written February 2004.
For more Arizona Production Ag Information:
Home | Cotton | Veggies| Forages | Grains | Citrus | Crop x Crop | Insects | Diseases| Weeds | Pesticides | News | Weather | Research | Photos | Contacts | General Info. | Site Map
document located at: http://cals.arizona.edu/crops/counties/yuma/farmnotes/2004/fn0204foxglove.html
Copyright © 2001 University of Arizona,
College of Agriculture and Life Sciences
Webmaster: Al Fournier (fournier@ag.arizona.edu)