Cotton IPM in Arizona: A Decade of Research, Implementation
& Education
Peter C. Ellsworth & Jennifer S. Jones
University of Arizona, Maricopa, AZ
Pest management in Arizona during the 1990’s has been filled with unique challenges and solutions. This tumultuous period was dotted with pest outbreaks, grower adaptation and perseverance, and spectacular recovery and progress. The elements that helped broker these tremendous successes cotton grow-ers now enjoy—at least with respect to pest management— were comprised of multiple groups who worked individually and together towards solutions. The University of Arizona, the Land Grant University in this state, and Cooperative Extension have been at the center of response to many of these imposing challenges to this important industry. This paper briefly details a historical review of the pest pressures and the specific response to one pest in terms of research, implementation, and education. The importance of our mission as a Land Grant institution has been instrumental in catalyzing a multilateral response to and recovery from insect pest pressures that would most assuredly have been fatal to the cotton industry of this state.
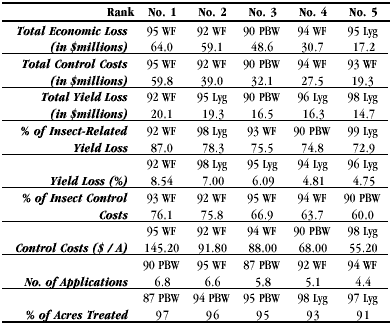
Arizona’s pest complex has shifted over time due to natural and man-influenced factors that are not entirely understood. Throughout the last decade, however, the primary pests of concern have not changed: Pink Bollworm (PBW), Bemisia whiteflies (SWF), and Lygus bugs. These three pests dominate the attention and pest control budgets of our growers. In an analysis of pest control trends over the past 22 years (Table 1), only two occurrences outside of the 1990’s appeared in the rankings of severe pest incidences (‘years’). Also, no pest other than these three primary pests have ever ranked in the top five most severe pest incidences over nine categories of classification (Table 1: 11 for PBW; 13 for Lygus; 21 for SWF).
Table 1: Ranking of top 5 pest-years for each insect loss statistic from the last 22 years (data from Ellsworth & Jones, 2000). Notes: Only foliar insecticide and application costs are included. ## = 19##, the year of production; WF = Bemisia whiteflies; PBW = Pink Bollworm; Lyg = Lygus bugs.
Whiteflies
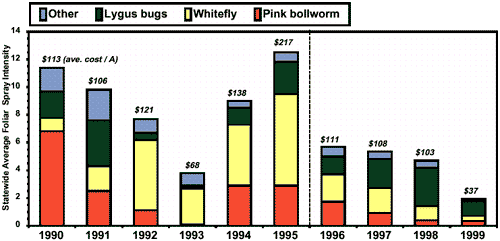
The 1992 SWF outbreak was the second costliest on record and impacted yields more than any other pest (Table 1). Vast areas of cotton plantings were biologically defoliated due to the stress imposed by this insect. By 1993, growers adopted a defensive production strategy that included a much abbreviated season, and this along with other advances in understanding helped to limit the damage by this pest but not without its cost. The largest portion in history of a grower’s control budget was spent fighting this pest in that year (Table 1; Fig. 1). The following year (1994) continued a trend of increasing costs to control this pest ($/A; 3rd most expensive on record). But by 1995, SWF became the most costly outbreak of a pest in AZ cotton in history. Foliar spray intensity was higher in 1995 than in any other year of the 1990’s and in history (Fig. 1).
Figure 1: Statewide average foliar insecticide intensity
statistics for Arizona cotton: number of foliar sprays by pest (bars)
and average costs per acre (including applications; above). Foliar sprays
are more a measure of insecticide use intensity than literal passes or
applications over fields. Bt transgenic cotton effective against pink
bollworms, insect growth regulators effective against whiteflies, and
a new IPM plan and educational campaign were introduced in 1996 (dashed
line) (see Fig. 2).
Altogether, the SWF infestations of 1992–1995 dominate the rankings of pest severity (Table 1) and occupy the No. 1 position in 7 of 9 categories of impact. SWF deposits honeydew, a viscous sugary excrement, that contaminates lint and prevents efficient processing. The costs of these quality losses to the industry or the reductions in price to growers for their cotton in this state during this period were not included in any of these analyses. Thus, SWF’s economic and historic importance to AZ cotton is even greater than reported here. SWF remains a key pest of our system; however, no occurrences of this pest ranked in the top 5 most severe for the variables measured since 1995 (Table 1).
The Sweetpotato Whitefly [Bemisia tabaci Genn. (Strain B) = Bemisia argentifolii Bellows & Perring (Silverleaf Whitefly)] was likely introduced into Arizona in the late 1980’s. During the 1990’s in Arizona, SWF rapidly supplanted the old strain of Bemisia tabaci which rarely required control. It spread generally throughout most of Arizona in 1991 and broke out in unprecedented numbers in 1992. Whiteflies of both strains were likely present in various portions of Arizona in 1990. By 1991, SWF was well-established in Yuma County, AZ, season-long, and confined mainly to the late season in central AZ. Since 1992, SWF has been widely distributed throughout the cotton growing regions of AZ with the exception of Cochise and Greenlee Counties at our highest cotton elevations. SWF develops earliest (June) at the warmest, lowest elevation locations of Yuma County (nr sea-level). Arizona’s central production region experiences initial threshold level populations of SWF during July or August (elev. ca. 1000 ft). At 2000 ft elevation, SWF occasionally reach threshold levels (Pima & portions of Maricopa Counties). The Safford Valley of Graham County is the highest elevation at which threshold level populations are possible, albeit infrequently (ca. 3000 ft). SWF is the largest destructive force on Arizona’s cotton industry than any other pest. Besides limiting the length of the cotton season during the early 1990’s, certain fall crops were curtailed altogether. Cotton production [1991–1992] was reduced in the Mexicali Valley (in Mexico, adjacent to Yuma and Imperial Counties) by over 98% mainly due to the ravages of this pest. This event was a harbinger of what was to come to central AZ in 1992. The 1992 outbreak was most certainly related, at least in part, to a lack of readiness on the part of the cotton industry including the research/extension community. Little was known about the manner in which to sample or control this insect nor about its prodigious ability to overcome insecticides.
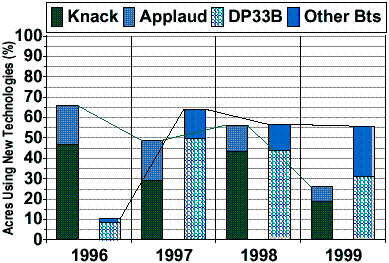
Figure 2: Estimates of adoption rates for new pest management technologies in Arizona cotton. The IGRs, Knack and Applaud, selective against whiteflies were made available under Section 18 Emergency Exemption starting in 1996. Transgenic Bt cottons were commercially introduced in 1996 with limited availability in limited varietal backgrounds. DP33B dominated Bt varieties planted in Arizona. Actual adoption is likely slightly higher (ca. +6%) than reported here (ave. approx. 64% since 1997)
Research
After a rapid expansion of range from Florida to California during the 1980’s, Arizona was subject to some of the first chronic economic infestations of SWF in cotton in U.S. history. Early research [1991–1993] quickly identified the necessity for mixing pyrethroids with organophosphate or other synergists. Pyrethroids or other compounds used alone provided little or no protection against this pest. In spite of this pest’s worldwide significance, a systematic sampling plan for cotton had not been developed. Field studies [1993–1994] by the USDA-ARS described immature and adult distributions on cotton useful for designing sampling plans. With these tools for sampling in hand, action levels were first tested and defined [1993], leading to a multi-state, multi-agency program of thresholds testing [1994–1996]. Economic injury levels were also defined for the first time in cotton. Later, field-testing of novel Insect Growth Regulators (IGRs: Knack ® andApplaud ® ) lead to their landmark emergency registration in 1996. Research into resistance mechanisms, resistance monitoring, andresistance management helped to understand the refractory nature of SWF resistances and the need for proactive management to preserve susceptibilities for the future [1991–]. Recent studies [1996–] have begun to identify the factors which make our current system of management so successful, and may chart a path toward even greater gains in natural enemy conservation and other non-intrusive means of ecological management of SWF.
Implementation
Growers organized community IPM programs, but were quickly challenged by resistances in SWF to conventional chemistry [1993–1996]. These programs include coordinated sampling and sharing of information that helped both individuals and collectives make strategic decisions about insecticide use.Implementation of adult sampling procedures within an 18,000 A community lead to commercial validation of this critical IPM tool [1993–1994]. Proper rotations of conventional chemistry and use of sampling and moderate thresholds were implemented at an unprecedented scale (ca. 210 A) in a multi-agency, commercial-scale experiment [1995] (Fig. 3). After the 1995 outbreak, a multi-agency and industry coalition developed a new strategy that included the use of two unregistered, whitefly-specific IGRs. An unprecedented Section 18 Emergency Exemption was granted by EPA [1996–] for both compounds under a strict plan of one-use each, user certification, and mandatory education. These IGRs with new sampling procedures and action thresholds were subsequently tested in a multi-agency collaboration [1996] (Fig. 4) and simultaneously implemented on over 13,000 A of commercial cotton farms. Large-scale, demonstrations of IGR’s were integrated with other best management practices [1998–2000]. IGR adoption and use was immediate and dramatic with two thirds of the acres using IGR’s in 1996 (Fig. 2). Usage has since declined, but oddly, this is due to increased producer confidence in the SWF control tools at their disposal. Other factors include a generalized decline in severity of ambient populations or areawide suppression (e.g., Fig. 4).
Education
Early educational efforts focussed on instilling understanding in growers of the life cycle, biology, and seasonal ecology of this pest. Training sessions were held for Cooperative Extension personnel, Master Gardeners, producers and others [1994]. A multi-regional manual on the biology, ecology and control of this pest across multiple crops was produced and distributed in four states. Community action plans were developed and disseminated within grower groups interested in collective management of this pest. Early research into the relative efficacy of insecticidal mixtures and a rudimentary rotational scheme was extended to growers via a laminated pocket guide and brochure. This popular guide was updated to include the IGRs and distributed to hundreds of growers in AZ and translated into Spanish and distributed in N. Mexico. A multilateral, grower-endorsed and -imposed, mandated educational campaign was launched [1996–]. The focus was stewardship of first-ever, field crop use of IGRs through 1) proper sampling and action thresholds including adults and nymphs, and 2) an aggressive, proactive resistance management program that includes 1 use per season IGR limits as part of an overall IPM approach. Over 700 PCAs were trained and certified for proper use of IGRs. Growers were taught about the lack of adult knockdown with these new products and the need to wait at least 7–10 d after spraying IGRs before observing significant nymphal mortality. Growers were mandated by Section 18 label to wait a minimum period after the use of one IGR before applying the alternate IGR if necessary. A sampling count card was produced and thousands distributed with a fibrous washer that identified the proper location and area for counting SWF large nymphs. A dual-component binomial sampling and threshold system was taught to hundreds of PCAs and growers. Continued efforts today emphasize cross-commodity cooperation and management of SWF in a sustainable manner.
“What if…?”
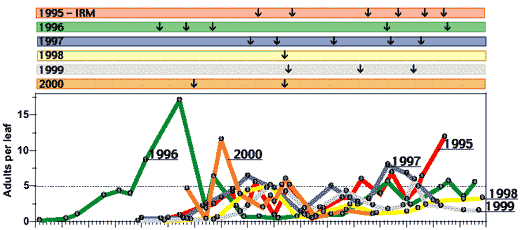
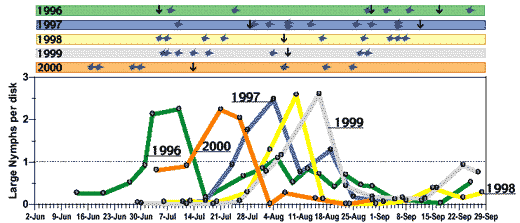
As with any historical account, it becomes difficult to state with certainty causation, especially with respect to biological phenomena such as insect infestations or outbreaks. As a result, it begs the question, “What if…?” What if certain practices or technologies or teachings had not occurred? Would the course of pest management and cotton production been different? Undoubtedly some things would be different, but it is difficult to establish alternative scenarios with any degree of certainty. However, in at least one case (SWF), we can get a comparative glimpse into potential alternative outcomes through a series of large-scale, systematically replicated trials (Fig. 3–4).
Whitefly population dynamics have been systematically studied on large, commercial-scale, yet experimental, acreages since 1995 prior to the introduction of the Insect Growth Regulators (IGRs). At that time, our best management practices included a rotation of non-pyrethroid and pyrethroid combinations (‘95IRM’) applied on an adult threshold of 5 adults / leaf (the University recommendations). This approach has been maintained in a comparative manner in replicated large-scale plots through 1999. In 2000, fiscal constraints prevented true replication; however, one 21-A field was divided into thirds that were assigned to one of three regimes (IGRs, conventional chemistry, and an untreated check). All other pests were managed normally—pink bollworm was controlled by the Bt transgenic variety (DP33B except in 2000, DP458B/RR) and Lygus bugs were sprayed as needed with single compounds (usually once or twice in July).
The results allow us to make comparative observations over this 6-year period (1995–2000). As seen by Figure 3, adult levels were similar among years with few exceptions. In 1996, adults reached threshold sooner than in any other year requiring a total of 5 conventional sprays. In 1995, the last severe outbreak year, atypical adult dynamics were not seen until September when populations exploded in spite of 6 conventional sprays. In 2000, the second earliest onset of threshold levels was observed; however, a combination of rains, time, and other weather factors probably mitigated any potential for outbreak levels of whiteflies or stickiness.
Since 1997, whiteflies have been successfully managed with this conventional rotation using only 1–3 sprays. This past year (2000) might have required more than the 2 sprays used, if it had not been for the rather active monsoon season that contributed to dramatic reductions in whitefly populations. Putanother way, had there been a weak monsoon with very dry weather as was more typical in the earlier 1990’s, 4 or 5 conventional sprays might have been required in 2000. In general, however, 1995–1997 required 5–6 conventional sprays, while 1998–2000 required only 1–3 conventional sprays. The specific reason for these differences is under investigation but likely was a result of weather, predation, and unknown factors as well as the surrounding ambient density of SWF.
The IGR regime performed exceptionally well in all years requiring usually just 1 spray to manage SWF season-long (Fig. 4). The IGR regime required on average 1–3 fewer sprays against SWF than did the conventional regime. Only in 1998 were the spray requirements the same (1). The SWF dynamics in this year were extraordinary in that threshold levels were present statewide during the first week of August, but then spontaneously crashed even in untreated plots and commercial fields. As measured by large nymph densities (Fig. 4), threshold levels of SWF were reached at distinctly differenttimes each year. Due to the time lag effects inherent to IGR’s, the peak nymphal densities were always reached about 1 week after IGR use. The characteristic precipitous decline thereafter was often followed by 3–7 weeks of subthreshold SWF densities.
This historical comparison is useful in re-evaluating University recommendations within a commercial-scale context and for revisiting the relative utility of conventional chemistry vs. IGRs, a choice growers face each year. Also, this comparison provides a useful ‘what if’ situation where we can see the importance of properly deployed IGRs in combatting SWF more economically and more eco-rationally. Furthermore, it is apparent from this exercise that this integrated use of IGRs and all the associated research, implementation, and education are likely mostly responsible for the staggering reductions in insecticide use against SWF in Arizona cotton. This conclusion is supported by independent analyses of pesticide use trends.
Figure 3 & 4: Historical trends in whitefly populations dynamics (adults per leaf & large nymphs per disk) and conventional (top) & IGR (bottom) spray requirements from large-scale, replicated experiments. The same sequence of conventional insecticides (top) was used in each year according to a rotational regime identified in 1995 for resistance management. Insect growth regulators (IGRs) were used before conventional chemistry in each year (data for Knack first shown only; bottom). Arrows above chart indicate frequency and timing of sprays by year. Numbered points on lines correspond to the last digit of the year (e.g., ‘8’ for 1998). Cloud-bursts above chart denote rain events for each year. (higher resolution PDF version, 50KB of Figs. 3 & 4)
Issued in furtherance of Cooperative Extension work, acts of May 8 and June 30, 1914, in cooperation with the U.S. Department of Agriculture, James A. Christenson, Director Cooperative Extension, College of Agriculture and Life Sciences, The University of Arizona.
The University of Arizona is an equal opportunity, affirmative action institution. The University does not discriminate on the basis of race, color, religion, sex, national origin, age, disability, veteran status, or sexual orientation in its programs and activities.
Any products, services, or organizations that are
mentioned, shown, or indirectly implied in this web document do not imply
endorsement by The University of Arizona.
Information provided by:
Peter C. Ellsworth, peterell@ag.arizona.edu Specialist, IPM/Entomology
University of Arizona, Tucson, Arizona.
Material written April 2001.
For more Arizona Production Ag Information:
Home | Cotton | Veggies| Forages | Grains | Citrus | Crop x Crop | Insects | Diseases| Weeds | Pesticides | News | Weather | Research | Photos | Contacts | General Info. | Site Map
document located at: http://cals.arizona.edu/crops/counties/yuma/farmnotes/fn0401cottonIPM.html
Copyright © 2001 University of Arizona,
College of Agriculture and Life Sciences
Webmaster: Al Fournier (fournier@ag.arizona.edu)